Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
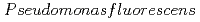
Nankawa, Takuya; Suzuki, Yoshinori*; Ozaki, Takuo; Onuki, Toshihiko; Francis, A. J.*
Journal of Alloys and Compounds, 408-412, p.1329 - 1333, 2006/02
Times Cited Count:3 Percentile:28.65(Chemistry, Physical)We studied the biodegradation of Eu(III)-malic acid complexes by 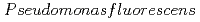 . Ten milimolar Malic acid was degraded in the absence and in the presence of Eu(III) of 0.05, 0.1, and 0.2 mM. The degradation rate of malic acid increased with decreasing the ratios of Eu(III) to malic acid. These results suggest that the toxicity of Eu(III) can be masked through its complexation with malic acid. The degradation of malic acid was followed by the production of unidentified metabolites which were associated with Eu(III). One of the unidentified organic acids was analysed to be pyruvic acid. Our findings suggest that metabolites can influence the environmental behavior of Eu(III) by biologically transformed through subsequent complexation with Eu(III).
. Ten milimolar Malic acid was degraded in the absence and in the presence of Eu(III) of 0.05, 0.1, and 0.2 mM. The degradation rate of malic acid increased with decreasing the ratios of Eu(III) to malic acid. These results suggest that the toxicity of Eu(III) can be masked through its complexation with malic acid. The degradation of malic acid was followed by the production of unidentified metabolites which were associated with Eu(III). One of the unidentified organic acids was analysed to be pyruvic acid. Our findings suggest that metabolites can influence the environmental behavior of Eu(III) by biologically transformed through subsequent complexation with Eu(III).